Site Index
- Home
-
Research - Background Information
-
Research - Mobility of Taxol in Tight Microtubule Bundles
-
Research - Diffusion of Macromolecules Inside Microtubules
-
Research - Tau Induces Cooperative Taxol Binding to Microtubules
-
Publications and Presentations
- Classes
Links
Research
Tau Induces Taxol to Bind Microtubules Cooperatively
Taxol and tau both stabilize and promote assembly of the labile microtubule lattice by binding to microtubules. Taxol is a small cancer therapeutic that binding to b tubulin on the interior surface of the microtubule. Tau is a microtubule-associated protein (MAP) found in the axons of neuronal cells. It is a natively unfolded protein that strengthens and spaces microtubules when it binds to the exterior of microtubules in vivo. Tau and taxol were recently shown to interact by cryo-EM reconstructive imaging (Kar, et. al. FEBS 2003). This study observed tau binding to the taxol binding site on the interior of microtubule. In order to assess this possible interaction, we use fluorescence recovery after photobleaching (FRAP) using both a fluorescently-labeled taxol and tau. See the page on taxol FRAP for information on the method of FRAP.
Dissociation of Tau from Microtubules We found that recovery of fluorescent tau was unaffected by the presence of taxol or the microtubule assembly conditions (with or without tau). Recovery of fluorescent tau had two recovery times - one fast and one slow. The fast recovery time is due to diffusion of unbound tau around the microtubules, and is similar to the diffusion of a similarly sized dextran (70 kD) around tau-coated microtubules. The long recovery time for tau, 2500 sec, was longer than any seen for dextran, which was complete within 500 seconds. This slow recovery time represents dissociation of the tau from the microtubule substrate, and gives us an estimate for the dissociation rate of tau: koff~1/(42 min). Since the presence of taxol and assembly conditions produce the same recovery curves, we infer that the off rate is unaffected by assembly conditions.
Tau Induces Taxol to Bind Cooperatively As in our previous study, we find the recovery time for taxol binding depends on the taxol concentration in free solution (see taxol FRAP). At low tau concentration (0 and 1:50 tau:tubulin molar ratio), the recovery time as a function of taxol concentration is the same as the previous study. We find the recovery time is a sigmoid with a Hill coefficient nH = 1, indicating no cooperativity in taxol binding. At higher tau concentrations, 1:10 and 1:1 tau:tubulin, the recovery times become more sensitive to the free taxol concentration, reaching a Hill coefficient of 15 at the highest tau concentrations. Hill coefficients greater than one signify a cooperative reaction. Cooperative binding means that several taxol molecules like to bind and unbind at the same time. The Hill coefficient is an estimate of the number of taxols that are binding cooperatively. Since the taxol molecules are not tethered together, the cooperativity must be caused by an increase in affinity for the binding site when a neighboring site is already bound with taxol. We observe that the taxol binds cooperatively when tau is bound to microtubules. The size of the cooperative interaction, as measured by the Hill coefficient, nH, depends on the amount of tau. Our study is the first to observe a possible cooperativity in taxol binding to microtubules.
|
FIGURE: The first column (I) is the data for the fluorescence recovery time as a function
of taxol concentration for 3 different concentrations of tau: A. 0 tau and 1 tau
for 50 tubulin dimers, B. 1 tau for 10 tubulin dimers, and C. 1 tau for 1 tubulin
dimer. The second column (II) is a cartoon depiction of the taxol binding reaction at each of the three tau concentrations. |
 |
|
A. At low tau concentration, the plot shows a gently rising curve. The Hill coefficient
of such a plot is 1, indicating no cooperativity in binding. This implies, as depicted in
the cartoon, that one taxol binds to one binding site and does not affect the binding of any
other taxol molecules.
|
 |
|
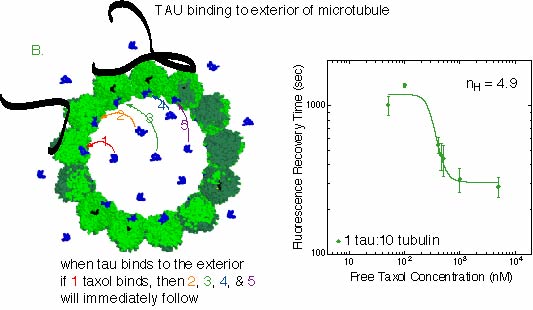
B. At intermediate tau concentration, the data is a similar curve, by the slope is
much steeper. The plot is flattened at high and low taxol concentrations because the
increased slope has caused these areas to be better defined. The Hill coefficient of this plot
is about 5, implying that 5 taxols bind to sites cooperatively. This means that 5 taxols
all bind at almost the same time, because the first one binding increases the probability that
the next one will bind, and so on until all 5 are bound. In order to unbind from the sies,
all 5 have to dissociate at the same time.
|
 |
|
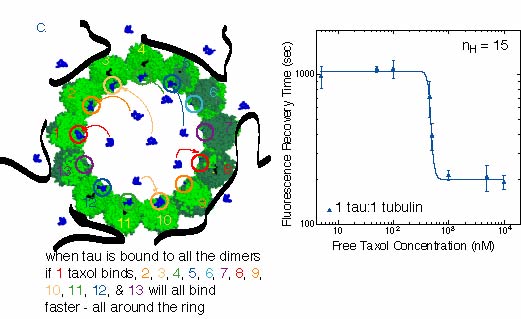
C. At the highest tau concentration, the microtubules should be saturated with
tau binding on the exterior, as shown in the picture. The data plot shows that the transition
in recovery times between low and high taxol concentrations is very steep. The
Hill coefficient is 15, which is a very high amount of cooperativity. Coincidentally,
15 is approximately the number of dimers around the microtubule ~12-15, pictured in the cartoon.
It could be that the exterior bound tau is causing all the taxol binding sites to bind
taxol cooperatively, as explained above. It is possible that the cooperativity only affects
the next taxol around the circle. The maximum number of affected sites around the ring is 12-15.
This is due to the structure of the microtubule lattice. Typically, dimers stack
near each other such that a is next to b
in the lattice. To have 15 dimers around causes the lattice to produce a seam
that causes an a to bind next to another
a instead of a b at the seam. The presence
of a seam in a 13-protofilament microtubule would cause a limit to the maximum Hill
coefficient,
even though all the taxol binding sites could behave cooperatively.
In order to understand the mechanisms for taxol binding, we have devised a model that accounts for the results of our study. Chris Santangelo has provided a theory that explains the (1) constant width of the gaussian intensity profiles over time, (2) exponential decay of the amplitude as a function of time, and (3) dependence of recovery time on free taxol concentration at low tau concentrations. Using both a master equation and a mean-field theory approach, we find that rebinding events at a single binding site and escape through pores in the microtubule wall dominate taxol binding visualized in the taxol FRAP experiments. For the abstract and a PDF version of this paper, please click: Publications
|

 Background Information
Background Information Mobility of Taxol in Tight Microtubule Bundles
Mobility of Taxol in Tight Microtubule Bundles Mobility in Tau-Coated Microtubule Bundles
Mobility in Tau-Coated Microtubule Bundles How Tau Affects Taxol Mobility
How Tau Affects Taxol Mobility